The Siemens Healthineers FAST III study has demonstrated that a less-invasive method for assessing coronary artery disease can deliver comparable outcomes to conventional techniques, supporting wider adoption of angiography-based diagnostics.
FAST III study validates non-invasive coronary assessment
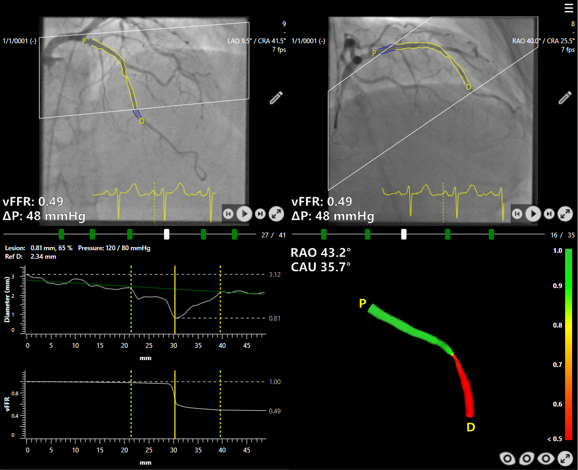
Results from the FAST III study, presented at the American College of Cardiology Congress and published in the New England Journal of Medicine, confirm that angiography-based vessel fractional flow reserve (vFFR) can guide coronary interventions as effectively as traditional pressure wire-based methods.
The study evaluated more than 2,200 patients across 37 hospitals in Europe, comparing outcomes between vFFR-guided and pressure wire-guided treatment strategies.
Reducing the need for invasive procedures
The vFFR approach uses software to calculate blood flow restriction from angiographic images, removing the need for pressure wires and pharmacologically induced hyperemia. This may reduce patient discomfort and procedural complexity.
According to the study findings, the method achieved comparable clinical outcomes, supporting its use as an alternative in patients with intermediate coronary artery stenosis.
Implications for cardiovascular care pathways
Coronary artery disease remains a leading cause of death globally, with accurate assessment of blood flow critical to determining appropriate treatment. Traditional methods require invasive instrumentation and can be associated with patient discomfort.
The FAST III results suggest that less-invasive approaches could streamline workflows, expand access to physiological testing, and support decision-making in centres performing diagnostic angiography.
Technology and collaboration underpin study
The study was sponsored by the European Cardiovascular Research Institute and co-funded by Siemens Healthineers and Pie Medical Imaging. The vFFR technology used in the study is based on software developed by Pie Medical Imaging.
Further information is available via the Siemens Healthineers website. Related coverage can be found in the Medical Devices & Diagnostics section.